Minimum daily PCR Cultures for lab to be a profitable business:
7 wound and/or urine cultures per day, you should be doing PCR cultures in-house.
7 toxicology samples being sent out/day
10-15/d: next gene sequencing biomarkers: alz, diabetes,
100 blood/day general labs: CBC, electrolytes ; multiple panels not just CBC ie hormone, cardiac panels
30-40 nails/day
Financial Benefits of Physician Owned Labs: PCR, Toxicology, Pharmacogenetics, Next Gene Sequencing
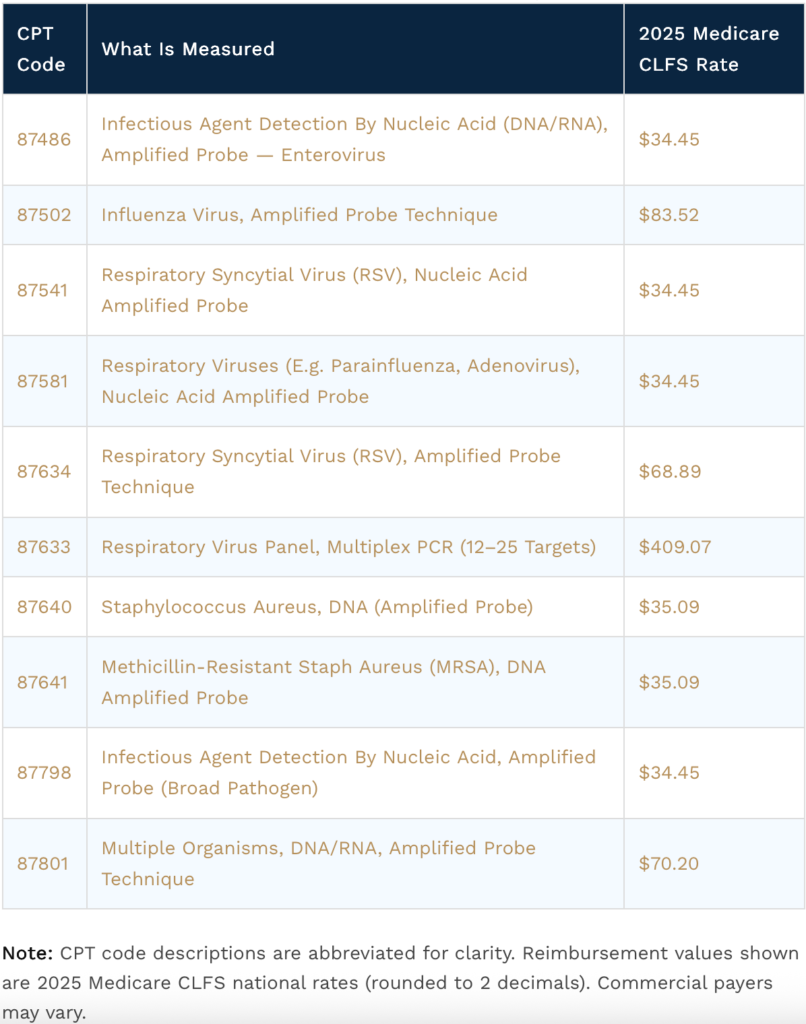
Medicare reimbursement ~$1,100 per PCR culture based on Medicare Fee Schedule:
Owned Lab Gross Based / Sample- based on # of targets but panels can be customized. Most common panels:
Hosted Lab Gross Based/Sample
- Urine $1100
- Wound $1100
- Toxicology $200;
- PCR: $200-$700;
- PGX: $350
- NGS (Next Gene Sequencing $450-$12,000
Example: 10 wound/urine PCR cultures/day →$ 11,000/Day
Hosted Lab ROI
Estimated average provider reimbursement per test for hosted lab models. Ranges vary by payer mix, volume, and regional MAC guidance.
| Test Category | Provider Net Revenue (per test) | Notes |
|---|---|---|
| Toxicology | ~$200 | Screen + confirmation workflows |
| PCR Testing | ~$200–$700 | Respiratory, GI, wound, UTI; fast TAT |
| Pharmacogenetic (PGx) Testing | ~$350 | Guides safer, effective prescribing |
| NGS (Next-Generation Sequencing) | ~$450–$12,000 | Targeted panels to comprehensive genomic assays |
| Comprehensive Wellness Panels (Blood) | ~$350 | Must include: CBC, A1C, CMP, RFP, Hormone panel, Vitamin panel, Serology panel |
Pharmacogenetics with NGS (SEO Callout)
Next-Generation Sequencing (NGS) enables pharmacogenetic testing by detecting common and rare variants in pharmacogenes, producing a complete pharmacogenomic profile that improves medication selection and outcomes.
Tests Typically Not Beneficial to Run In Physician-Owned Labs
When run as solo, unqualified blood draws (i.e., outside a comprehensive wellness panel), reimbursement is often lower than internal cost:
- CBC: reimburses ~$3
- BMP: reimburses ~$12
- CMP: reimburses ~$18
Recommendation: Bundle blood tests into comprehensive wellness panels to achieve better reimbursement and clinical value. For a practice-specific analysis, request a custom pro forma.
Out of state labs not allowed
Out of state labs not allowed: NY, NJ, RI CT, WA, CA, WA=Dc
Lab Setup Requirements
Central processing lab for couriered/overnighted samples
Space requirements:
- 250/day → ~120 sq ft
- 2000/day → ~500 sq ft
- 3000/day → ~750 sq ft
Timeline: 6–8 weeks to full go-live for in-office PCR cultures.
Our Turn-Key Value Proposition
- Obtain & submit High Complexity License (4–8 weeks)
- Maintain licensure, COLA follow-up
- Identify & train lab personnel: Director, Supervisors, Techs
- Provide & install all equipment & supplies
- Train staff on coding, billing, and PCR workflow
- Onsite live virus validation
- EHR integration with robust, cost-effective LIS
Equipment & Validation
- BioRad CFX96 PCR Machine (1-year warranty)
- Laptop, freezer, pipettes, centrifuge, heat blocks, swabs, proprietary reagents
- 5-day onsite installation + validation
- Validation includes specificity, sensitivity, stability studies → >99% accuracy
Panels & Performance
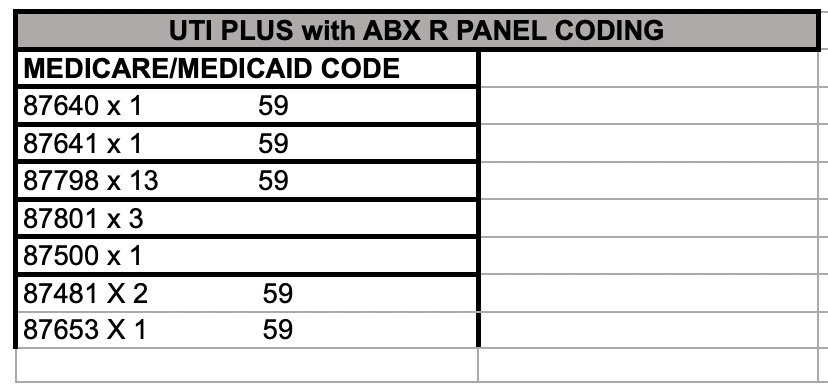
- Comprehensive Panels (UTI, Wound, Respiratory, Vaginitis, Ear, COVID) → higher reimbursement
- Proprietary reagents validated onsite
- One platform runs all PCR cultures
- Panels include antibiotic resistance markers
Reimbursement Examples:
UTI & Wound + ABX panel: ~$1,100/test (Medicare)
Investment & Ongoing Costs
- Eligible for Section 179 Deduction
- $65,000-85,000: Complete setup (all tests)
- Ongoing Monthly Costs (~$9K):
- Personnel/licensure fees
- High Complexity Lab Director (~$2.5–3K/month remote)
- Supervisors & techs (1 per 20 samples/day)
- CLIA fees ~$2,000/3 years
- Reagents: $/panel (UTI, Vaginitis, RPP, Ear, Wound, Covid)
Next Steps
- Schedule a Zoom call with Dr. O’Sullivan to review logistics, space, and staffing
- Receive a customized Pro Forma
- Sign agreement
- Go live in 6–8 weeks
